Prof. Dr. Bodo Grimbacher (CIBSS-AI), CCI Center for Chronic Immunodeficiency (University Medical Center Freiburg, Faculty of Medicine)
Prof. Dr. Barbara Di Ventura (CIBSS-AI), Institute of Biology III (Faculty of Biology), University of Freiburg
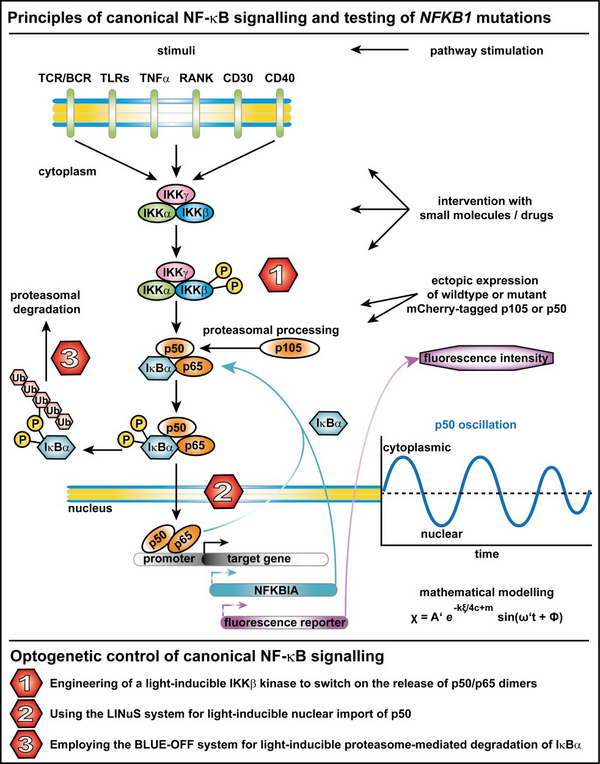
The highly complex NF-kB signalling network integrates signals from receptors, co-stimulators and metabolites in most cell types. Degradation of IkBα (the cytosolic inhibitor of NF-kB1) triggered by an upstream kinase (IKK), activates the dimeric NF-kB1 transcription factor (canonical pathway), which enters the nucleus and regulates its target genes, including IkBα itself. The transcriptional activation of its own inhibitor establishes a negative feedback loop that leads to NF-kB1 oscillations. In numerous patients with primary antibody deficiencies (PAD), we have identified heterozygous mutations in NFKB1, the gene encoding the p105 precursor, which is then further processed into the mature DNA binding subunit of the NF-kB1 complex (p50). The disease-causing mutations are predicted to give rise to inappropriate p105/p50 ratios as well as to impaired NF-kB1 dynamics.
We aim at a detailed analysis of the spatio-temporal regulation of the canonical NF-kB signal oscillation (including mathematical modelling), at identifying molecular switches (e.g. small molecules) to manipulate NF-kB activity in order to develop targeted therapies for human disease and at establishing optogenetic tools to control NF-kB1 signalling and its target gene expression.